Alcohol Metabolism

Giving the liver enough time to fully metabolize the ingested is the only effective way to avoid alcohol toxicity. Drinking coffee or taking a shower will not help. The legal limit for intoxication is a BAC of 0.08. Taking into account the rate at which the liver metabolizes alcohol after drinking stops, and the alcohol excretion rate, it takes at least five hours for a legally intoxicated person to achieve sobriety.
Ethanol Consumption
Distilled spirits have exceptionally few nutrients, but beer and wine do provide some nutrients, vitamins, , and beneficial plant chemicals along with calories. A typical beer is 150 kilocalories, a glass of wine contains approximately 80 kilocalories, and an ounce of hard liquor (without mixer) is around 65 kilocalories.
As a person starts drinking alcohol, up to 5% of the ingested ethanol is directly absorbed and metabolized by some of cells of the gastrointestinal tract (the mouth, tongue, esophagus and stomach). Up to 100% of the remaining ethanol travels in circulation. This is one reason why blood tests are more accurate in measuring alcohol levels.
The lungs and s will excrete about 2% to 10% of this circulatory ethanol. The more you drink the more quick trips to the restroom. The human body dehydrates as a result of these frequent trips to the restroom. This dehydration affects every single cell in your body, including your brain cells. This is the cause of the so-called “morning hangover”. Do not take Tylenol (acetaminophen). Alcohol metabolism activates an enzyme that transforms acetaminophen into a toxic metabolite that causes liver inflammation and damage. Liver damage may not be irreversible. Instead, drink water with or sport drinks to rehydrate the body’s cells.
Alcohol is a volatile (flammable) organic substance and can be converted to a gas. The lungs exhale alcohol as a gas. The more alcohol consumed, the stronger the smell of alcohol in a person’s breathe. Breathalyzer tests measure the exhaled alcohol levels in the lungs to determine the state of inebriation.
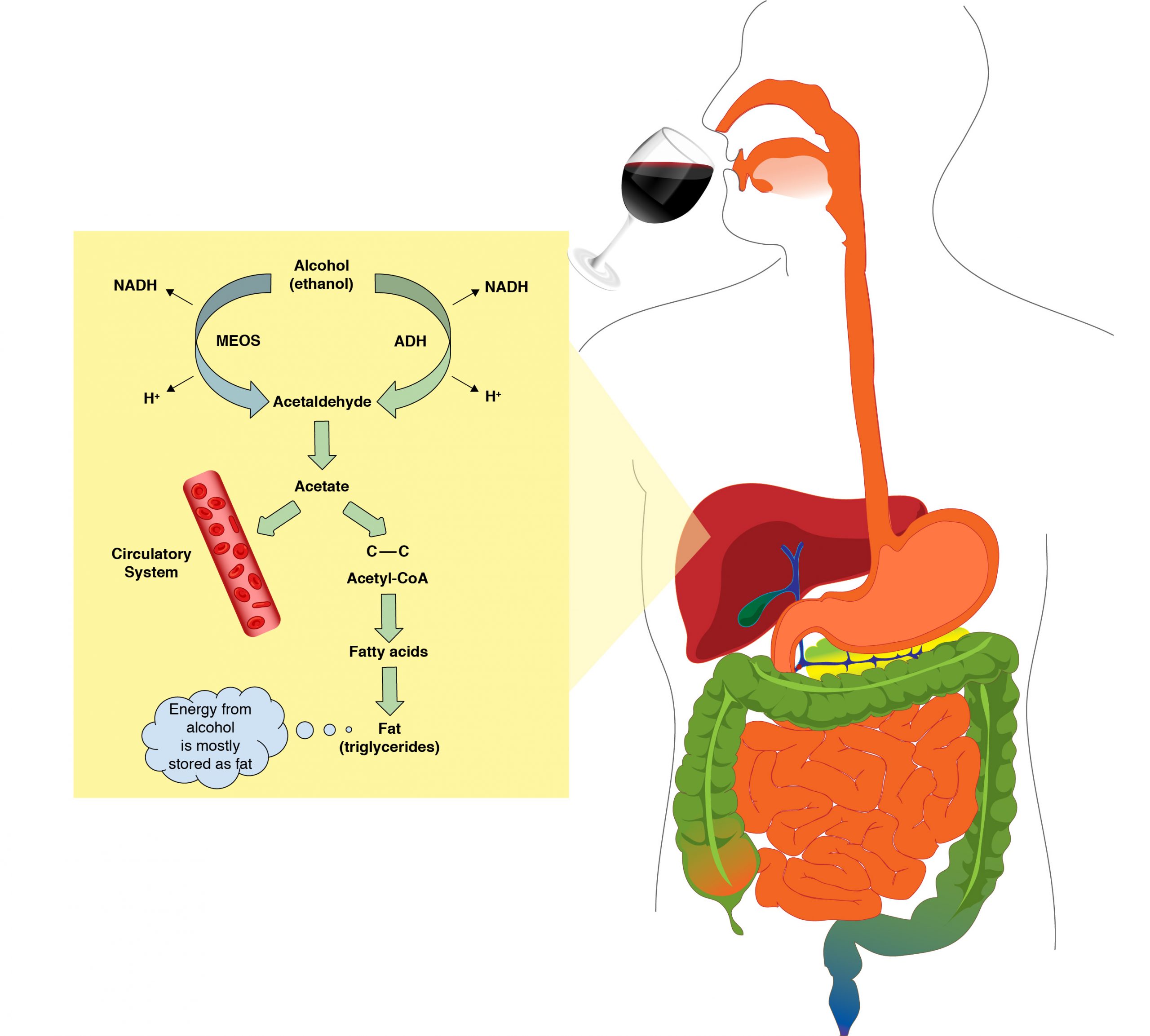
The liver metabolizes up to 85% – 98% of the circulatory ethanol. The liver uses two metabolic processes to get rid of this circulatory ethanol as quickly and safely as possible.
- system
- (MEOS)
Alcohol Dehydrogenase System
About 80 to 90% of the total hepatic ethanol uptake is processed via the alcohol dehydrogenase system. The degradation of ethanol begins in the liver. The enzyme that catalyzes this reaction is called alcohol dehydrogenase. The products from this reaction are acetaldehyde, NADH (a reduced coenzyme that carries from one reaction to another) and H+ ion. Acetaldehyde is very toxic to the liver and the body’s cells. The moment acetaldehyde is produced; it must be degraded to protect the liver cells. The enzyme that will carry this type of degradation reaction is acetaldehyde dehydrogenase (ALDH). Acetaldehyde dehydrogenase converts acetaldehyde into acetate, a non-toxic molecule.
Microsomal Ethanol Oxidizing System (MEOS)
In a moderate drinker, about 10 to 20% of the total liver ethanol uptake is processed via the microsomal ethanol oxidizing system (MEOS). During periods of heavy drinking, the MEOS system will metabolize most of the excess ethanol ingested. Heavy drinking stimulates the human body to include the MEOS system enzymes to clear ethanol faster from the body.
The MEOS system is also located in the liver. Similar to the Alcohol dehydrogenase system, acetaldehyde dehydrogenase will immediately convert acetaldehyde into acetate, a non-toxic molecule. Other products from this reaction are NADH and H+ ion.
Fate of Acetate
The acetate produced (from the alcohol dehydrogenase system and microsomal ethanol oxidizing system) is either released into circulation or retained inside the liver cells. In the liver cells, acetate is converted to where it is used to produce other molecules like CO2 or used in the synthesis of fatty acids and cholesterol.
Learning Activities
Technology Note: The second edition of the Human Nutrition Open Educational Resource (OER) textbook features interactive learning activities. These activities are available in the web-based textbook and not available in the downloadable versions (EPUB, Digital PDF, Print_PDF, or Open Document).
Learning activities may be used across various mobile devices, however, for the best user experience it is strongly recommended that users complete these activities using a desktop or laptop computer.
A molecule made from the fermentation of carbohydrates from plant products.
An element used in the body to promote chemical reactions and help form body structures.
A pair of organs that aid in filtering waste and water out of the blood while also maintaining chemical balance in our bodies.
Electrically charged ions dissolved in a solution. Commonly refers to sodium, potassium, and chloride.
An enzyme that breaks down ethanol and other alcohols into acetaldehyde in the first steps in the metabolism of alcohol in the liver.
Activated during periods of heavy drinking, this system metabolize excess ethanol ingested
Negatively charged particles found within the nuclei of atoms.
The acetylated form of coenzyme A that plays a central role in energy metabolism.
